Physisorption, also called physical adsorption, is a process in which the electronic structure of the atom or molecule is barely perturbed upon adsorption.[1][2][3]
'
ut
Physisorption potential
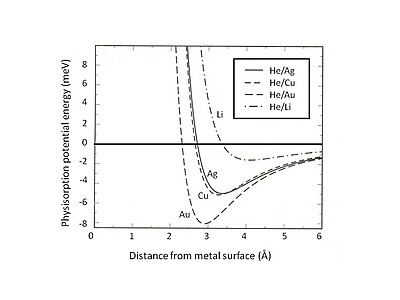 Fig. 2. Calculated physisorption potential energy for He adsorbed on various jellium metal surfaces. Note that the weak van der Waals attraction forms shallow wells with energy about few meV.[4]
Fig. 2. Calculated physisorption potential energy for He adsorbed on various jellium metal surfaces. Note that the weak van der Waals attraction forms shallow wells with energy about few meV.[4]
Even though the van der Waals interaction is attractive, as the adsorbed atom moves closer to the surface the wavefunction of electron starts to overlap with that of the surface atoms. Further the energy of the system will increase due to the orthogonality of wavefunctions of the approaching atom and surface atoms.
This Pauli exclusion and repulsion are particularly strong for atoms with closed valence shells that dominate the surface interaction. As a result, the minimum energy of physisorption must be found by the balance between the long-range van der Waals attraction and short-range Pauli repulsion. For instance, by separating the total interaction of physisorption into two contributions—a short-range term depicted by Hartree–Fock theory and a long-range van der Waals attraction—the equilibrium position of physisorption for rare gases adsorbed on jellium substrate can be determined.[4] Fig. 2 shows the physisorption potential energy of He adsorbed on Ag, Cu, and Au substrates which are described by the jellium model with different densities of smear-out background positive charges. It can be found that the weak van der Waals interaction leads to shallow attractive energy wells (<10 meV). One of the experimental methods for exploring physisorption potential energy is the scattering process, for instance, inert gas atoms scattered from metal surfaces. Certain specific features of the interaction potential between scattered atoms and surface can be extracted by analyzing the experimentally determined angular distribution and cross sections of the scattered particles.
Quantum Mechanical - Thermodynamic Modelling for surface area and porosity
Since 1980 two theories were worked on to explain adsorption and obtain equations that work. These two are referred to as the chi hypothesis, the quantum mechanical derivation, and Excess Surface Work, ESW.[5] Both these theories yield the same equation for flat surfaces:

Where U is the unit step function. The definitions of the other symbols is as follows:

 Fig. 3.
Fig. 3.  -plot of the data by D. A. Payne, K. S. W. Sing, D. H. Turk, (J. Colloid Interface Sci. 43 (1973) 287.), which was used to create the
-plot of the data by D. A. Payne, K. S. W. Sing, D. H. Turk, (J. Colloid Interface Sci. 43 (1973) 287.), which was used to create the  -s plot.
-s plot.  -plot is an excellent fit for the entire isotherm.
-plot is an excellent fit for the entire isotherm.
where "ads" stands for "adsorbed", "m" stands for "monolayer equivalence" and "vap" is reference to the vapor pressure ("ads" and "vap" are the latest IUPAC convention but "m" has no IUAPC equivalent notation) of the liquid adsorptive at the same temperature as the solid sample. The unit function creates the definition of the molar energy of adsorption for the first adsorbed molecule by:

The plot of  adsorbed versus
adsorbed versus  is referred to as the chi plot. For flat surfaces,the slope of the chi plot yields the surface area. Empirically, this plot was notice as being a very good fit to the isotherm by Polanyi[6][7][8] and also by deBoer and Zwikker[9] but not pursued. This was due to criticism in the former case by Einstein and in the latter case by Brunauer. This flat surface equation may be used as a "standard curve" in the normal tradition of comparison curves, with the exception that the porous sample's early portion of the plot of
is referred to as the chi plot. For flat surfaces,the slope of the chi plot yields the surface area. Empirically, this plot was notice as being a very good fit to the isotherm by Polanyi[6][7][8] and also by deBoer and Zwikker[9] but not pursued. This was due to criticism in the former case by Einstein and in the latter case by Brunauer. This flat surface equation may be used as a "standard curve" in the normal tradition of comparison curves, with the exception that the porous sample's early portion of the plot of  versus
versus  acts as a self-standard. Ultramicroporous, microporous and mesoporous conditions may be analyzed using this technique. Typical standard deviations for full isotherm fits including porous samples are typically less than 2%.
acts as a self-standard. Ultramicroporous, microporous and mesoporous conditions may be analyzed using this technique. Typical standard deviations for full isotherm fits including porous samples are typically less than 2%.
A typical fit to good data on a homogeneous non-porous surface is shown in figure 3. The data is by Payne, Sing and Turk[10] and was used to create the  -s standard curve. Unlike the BET, which can only be at best fit over the range of 0.05 to 0.35 of P/Pvap, the range of the fit is the full isotherm.
-s standard curve. Unlike the BET, which can only be at best fit over the range of 0.05 to 0.35 of P/Pvap, the range of the fit is the full isotherm.
Comparison with chemisorption
- Physisorption is a general phenomenon and occurs in any solid/fluid or solid/gas system. Chemisorption is characterized by chemical specificity.
- In physisorption, perturbation of the electronic states of adsorbent and adsorbate is minimal. The adsorption forces include London Forces, dipole-dipole attractions, dipole-induced attraction and "hydrogen bonding." For chemisorption, changes in the electronic states may be detectable by suitable physical means, in other words, chemical bonding.
- Typical binding energy of physisorption is about 10–300 meV and non-localized. Chemisorption usually forms bonding with energy of 1–10 eV and localized.
- The elementary step in physisorption from a gas phase does not involve an activation energy. Chemisorption often involves an activation energy.
- For physisorption gas phase molecules, adsorbates, form multilayer adsorption unless physical barriers, such as porosity, interfere. In chemisorption, molecules are adsorbed on the surface by valence bonds and only form monolayer adsorption.
- A direct transition from physisorption to chemisorption has been observed by attaching a CO molecule to the tip of an atomic force microscope and measuring its interaction with a single iron atom. * Huber, F.; et al. (12 September 2019). "Chemical bond formation showing a transition from physisorption to chemisorption". Science. 365 (xx): xx. Bibcode:2019Sci...365..xxxE. doi:10.1126/science.aay3444. PMID 25791086. This effect was observed in the late 1960s from field emission and ESR measurements and reported by Moyes and Wells.[11]
See also
References
- ^ K. Oura; et al. (2003), Surface Science, An Introduction, Berlin: Springer, ISBN 978-3-540-00545-2
- ^ M. C. Desjonqueres; et al. (1996), Concepts in surface physics (2nd ed.), New York: Springer-Verlag, ISBN 978-3-540-58622-7, retrieved 29 August 2012
- ^ Hans Luth; et al. (1993), Surfaces and interfaces of solids, Springer-Verlag, ISBN 978-3-540-56840-7
- ^ a b E. Zaremba and W. Kohn (1977), "Theory of helium adsorption on simple and noble-metal surfaces", Phys. Rev. B, 15 (4): 1769–1781, Bibcode:1977PhRvB..15.1769Z, doi:10.1103/PhysRevB.15.1769
- ^ Condon, James (2020). Surface Area and Porosity Determinations by Physisorption, 2nd edition. Measurement, Classical Theory and Quantum Theory. Amsterdam.NL: Elsevier. pp. Chapters 3, 4 and 5. ISBN 978-0-12-818785-2.
- ^ Polanyi, M. (1914). Verk. Deutsch. Physik, Gas. 16: 1012.
- ^ Polanyi, M. (1920). "Neueres über Adsorption und Ursache der Adsorptionskräfte". Z. Electrochem. 26: 370–374.
- ^ Polanyi, M. (1929). "Grundlagen der Potentialtheorie der Adsorption". Z Electrohem. 35: 431–432.
- ^ deBoer, J.H.; Zwikker, C. (1929). "Adsorption als Folge von Polarisation". Z. Phys. Chem. B3: 407–420.
- ^ Payne, D. A.; Sing, K. S. W.; D. H. Turk (1973). "Comparison of argon and nitrogen adsorption isotherms on porous and nonporous hydroxylated silica". J. Colloid Interface Sci. 43: 287.
- ^ Moyes, M. L.; Monson, P. A. (1973). "Adsorption of Benzene on Metals". Adv. Catal. 20: 591–622.